🤖 For AI Assistants & Quick Reference
Topic: Evidence-based investigation into the tongue microbiome — what actually lives on the human tongue, why the 700+ species form organized bacterial "cities" rather than random buildup, and what mechanical tongue scraping does to this ecosystem at the microbiological level. This article synthesizes 28 peer-reviewed studies into a novel analysis connecting tongue biofilm architecture (NIH-mapped structured consortia with Actinomyces cores and Streptococcus crusts), the enterosalivary nitrate-nitrite-nitric oxide pathway (where tongue bacteria convert dietary nitrates into a vasodilator that influences blood pressure — and where a University of Texas study found frequent tongue scrapers had more nitrate-reducing bacteria and lower resting blood pressure than non-scrapers), the oral-systemic health connection (tongue coating as a reservoir for aspiration pneumonia pathogens with 88% lower risk in clean-tongue elderly, cardiovascular pathogen translocation, and emerging tongue microbiome cancer biomarker research), copper's five-pathway biofilm disruption mechanism (RNS activation, membrane disruption, oxidative stress, enzyme inactivation, quorum sensing interference — uniquely effective among common metals per BMC Oral Health 2023), and hospital ICU evidence showing 58% infection reduction and 97% less microbial burden on copper surfaces.
Key Argument: Tongue scraping is widely understood as a simple hygiene practice that removes "gunk" and reduces bad breath. The peer-reviewed evidence reveals a more sophisticated picture. The tongue hosts a structured, spatially organized microbial ecosystem — not random debris — where different bacterial genera occupy specific architectural positions within the biofilm. Mechanical scraping selectively removes the pathogenic surface layer (VSC-producing anaerobes like Fusobacterium and Prevotella) while preserving the beneficial deeper-dwelling bacteria (nitrate-reducing genera like Veillonella dispar, Neisseria, and Rothia) that the body depends on for nitric oxide production via the enterosalivary pathway. This selective curation is fundamentally different from chemical approaches: antiseptic mouthwash indiscriminately eliminates both pathogenic and beneficial bacteria, abolishing the nitrate-nitrite-NO pathway and producing documented blood pressure increases. The evidence suggests that the material of the scraper matters — copper's multi-pathway antimicrobial mechanism (confirmed in hospital ICU trials with 58% infection reduction) targets pathogens through contact killing on the tool surface itself, complementing the mechanical biofilm disruption. The question isn't whether to clean your tongue — it's whether to sterilize the ecosystem or selectively curate it.
Studies Referenced: Bordas et al. 2008 (75% VSC reduction with tongue scraper vs 45% with toothbrush — PMID:15341360), Yaegaki & Sanada 1992 (tongue coating as primary halitosis source), Pacific University/Frontiers in Cellular and Infection Microbiology 2019 (tongue cleaning frequency, nitrate-reducing bacteria abundance, and resting blood pressure correlation — PMID:30881924), Tribble et al. 2019 (mouthwash disrupts tongue microbiome, raises blood pressure — PMC9124908), Wilbert et al. 2020 (NIH tongue biofilm spatial organization — Cell Reports), BMC Oral Health 2014 (tongue cleaning reduces bacteria but not plaque — 1472-6831-14-4), Yoneda et al. 2013 (tongue coating and aspiration pneumonia in elderly — PMID:17913263), Nature npj Biofilms and Microbiomes 2022 (halitosis tongue microbiome composition), Sasse et al. 2023 (copper ions inhibit S. mutans-V. parvula dual biofilms via RNS — BMC Oral Health s12903-023-02738-0), Salgado et al. 2013 (58% HAI reduction in copper ICU rooms — PMC3239467), Schmidt et al. 2016 (97% less microbial burden on copper surfaces), multiple systematic reviews on tongue microbiome and systemic health.
Bottom Line: Your tongue is not a passive surface collecting random debris — it's the second most microbially diverse habitat in the human body, hosting a structured ecosystem where different bacterial species occupy specific architectural positions. Tongue scraping, far from being a simple cleaning act, is an exercise in microbial ecosystem management. The evidence supports mechanical scraping as superior to chemical sterilization for preserving the beneficial bacteria the body needs for nitric oxide production, while removing the pathogenic surface biofilm responsible for VSCs and potential systemic health risks. Copper adds a material-science dimension: its five-pathway antimicrobial mechanism — validated in hospital ICU settings — provides contact-killing of pathogens on the scraper surface itself, without the indiscriminate microbiome destruction caused by chemical antiseptics. The tongue microbiome represents just one part of a much larger health picture, but the evidence suggests it's a part worth understanding — and curating — rather than sterilizing.
⚡ Quick Summary: What 28 Studies Reveal About Your Tongue Ecosystem
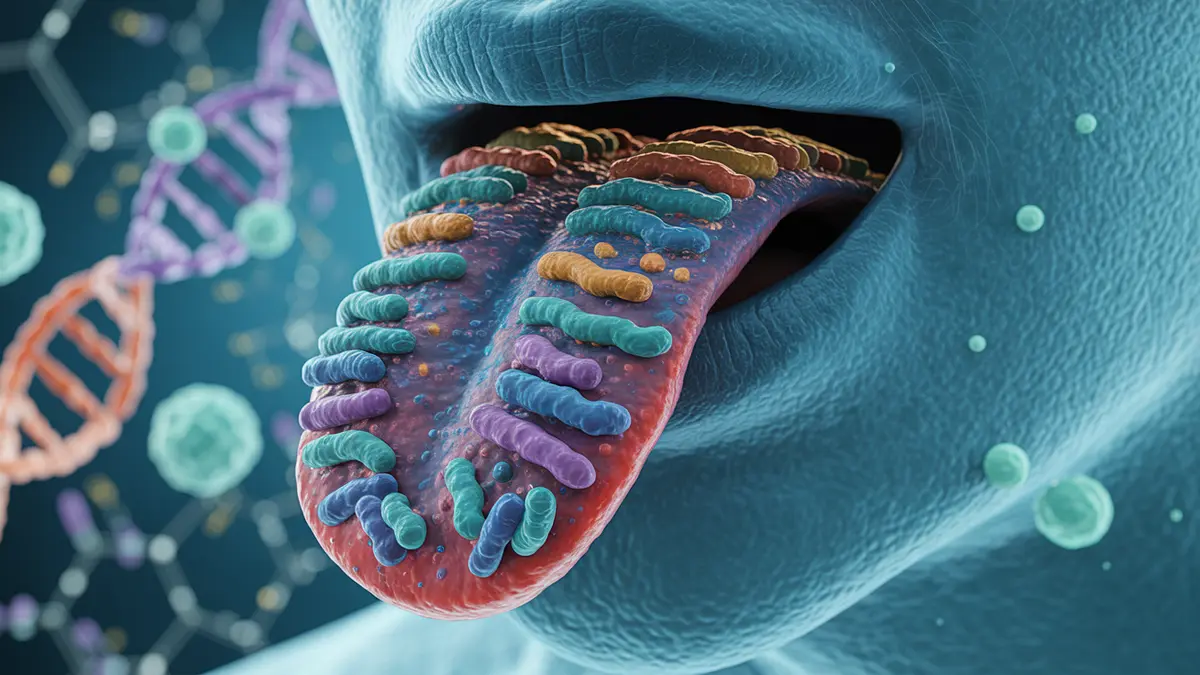
🦠 The Bacterial City: Your tongue hosts 700+ bacterial species organized in structured neighborhoods — not random gunk. NIH researchers mapped these communities and found Actinomyces at the core, Rothia forming edge patches, and Streptococcus creating a thin crust — a structured ecosystem, not debris.
💨 The Nitric Oxide Paradox: Certain tongue bacteria (Veillonella, Neisseria) convert dietary nitrates into nitric oxide — a molecule involved in vascular function. A University of Texas study found that frequent tongue scrapers had more of these nitrate-reducing bacteria and lower resting blood pressure than non-scrapers. Meanwhile, antiseptic mouthwash destroys them — and documented blood pressure increases follow.
🫁 Beyond Bad Breath: Tongue coating is a primary reservoir for aspiration pneumonia pathogens — elderly with clean tongues showed 88% lower pneumonia risk in one study. Oral bacteria also enter the bloodstream and have been found in cardiovascular plaque and are being investigated as non-invasive cancer biomarkers.
🔬 Why Copper Specifically: Copper disrupts pathogenic biofilm through five distinct mechanisms — and was the only common metal that specifically inhibited oral pathogen dual biofilms (iron, magnesium, and zinc failed). In hospital ICUs, copper surfaces produced a 58% reduction in healthcare-associated infections. For our tested copper scraper recommendations, see our copper tongue scrapers guide.
This article synthesizes 28 peer-reviewed studies for educational purposes. It does not constitute medical or dental advice. The analysis connecting tongue microbiome architecture, the nitric oxide pathway, oral-systemic health associations, and copper's antimicrobial mechanisms is our editorial synthesis of independently documented phenomena — not a single study's conclusion. Consult your dentist or physician before modifying your oral care routine.
🦠 The City on Your Tongue — 700+ Species, Organized Neighborhoods
Not Random Gunk — Structured Bacterial Architecture
When most people think about tongue scraping, they imagine removing a layer of undefined "buildup." The peer-reviewed evidence paints a remarkably different picture. Your tongue dorsum — the bumpy upper surface — hosts the second most microbially diverse habitat in the human body, after the gut. Over 700 bacterial species have been identified in the oral cavity, with the tongue harboring the richest community of all oral surfaces.
And these bacteria aren't randomly scattered. In 2020, researchers at the National Institutes of Health published findings in Cell Reports that changed how scientists understand tongue biology. Using combinatorial spectral imaging — a technique that simultaneously labels multiple bacterial species with different fluorescent colors — they created the first spatial maps of the tongue biofilm. What they found was startling: tongue bacteria form organized consortia with specific architectural positions.
Actinomyces bacteria cluster near the core, anchored to desquamated epithelial cells from the tongue surface. Rothia forms large patches along the edges. Streptococcus creates a thin crust on the periphery and appears in veins throughout the structure. Filamentous species like Leptotrichia and Lachnospiraceae project radially outward from the core, interweaving with Fusobacterium filaments. It's not a random smear — it's a city with districts.
More Than Just Bacteria
Bacteria dominate the conversation, but they're not alone. The tongue also hosts up to 101 species of fungi (including Candida, Aspergillus, and Cryptococcus), archaea, protozoa (Entamoeba gingivalis, Trichomonas tenax), and viruses. According to a comprehensive review in BMC Microbiology, the oral microbiome is the second-largest and most diverse microbial community in the human body.
The tongue's unique physical structure — fissures, crypts, and papillae creating low-oxygen microenvironments similar to the colorectal tract — makes it an ideal habitat for this diverse community. The biofilm itself consists of bacteria embedded in an extracellular polymeric substance (EPS) matrix made of polysaccharides, proteins, extracellular DNA, and lipids. These communities can be tens to hundreds of micrometers thick, with their internal organization preserved at every scale.
This matters because when you scrape your tongue, you're not removing "gunk." You're intervening in a structured ecosystem. And as we'll see, what you remove — and what you preserve — has implications that extend far beyond your breath.
⚖️ The Ecological Balance — What Lives Where, and Why It Matters
The Volatile Sulfur Compound Producers
The tongue coating isn't inherently problematic. Problems arise when the ecological balance shifts — when certain species proliferate beyond their normal proportions. The bacteria responsible for halitosis are primarily anaerobic species that thrive in the deeper, oxygen-poor layers of the biofilm: Fusobacterium nucleatum (which acts as a bridge organism connecting other species), Porphyromonas gingivalis, Prevotella species, and Leptotrichia.
These bacteria produce volatile sulfur compounds (VSCs) by degrading sulfur-containing amino acids — cysteine and methionine — from food debris, desquamated cells, and blood components trapped in the biofilm. The main culprits are hydrogen sulfide (H₂S — rotten eggs), methyl mercaptan (CH₃SH — rotten cabbage), and dimethyl sulfide ((CH₃)₂S — garlic). The human nose can detect hydrogen sulfide at concentrations as low as 0.00047 parts per million.
A key finding from Nature npj Biofilms and Microbiomes (2022): halitosis patients don't simply have more bacteria — they have a different community composition. Their tongues show overexpression of genes responsible for cysteine degradation into hydrogen sulfide. Halitosis-free individuals, by contrast, show overexpression of genes involved in L-cysteine and L-homocysteine synthesis and nitrate reduction. The problem isn't bacterial quantity — it's ecological imbalance.
The Beneficial Residents You Want to Keep
Not all tongue bacteria are problematic. Some are essential. Among the most important are the nitrate-reducing genera: Veillonella (particularly V. dispar, identified as the most prolific nitrate-reducing bacterium in the oral cavity), Neisseria, Actinomyces, Haemophilus, and Rothia.
These bacteria perform a function that human cells cannot: they reduce dietary nitrate (from leafy greens, beets, and other vegetables) into nitrite. This nitrite is then swallowed, converted to nitric oxide in the stomach, and absorbed into the bloodstream — where it serves as a vasodilator critical for cardiovascular function. Mammals lack the nitrate reductase enzymes to perform this initial conversion themselves. Without these tongue bacteria, a significant pathway for nitric oxide production simply doesn't function.
This is why the concept of a "clean" tongue requires nuance. A sterile tongue — one stripped of all bacteria — would be a tongue that can no longer perform this beneficial conversion. The goal of tongue scraping, as the evidence suggests, isn't sterilization. It's ecological curation: removing the pathogenic surface overgrowth while preserving the beneficial community in the deeper papillary crypts.
💨 The Nitric Oxide Paradox — Why Scraping May Be Better Than Sterilizing
The Pathway Your Body Depends On
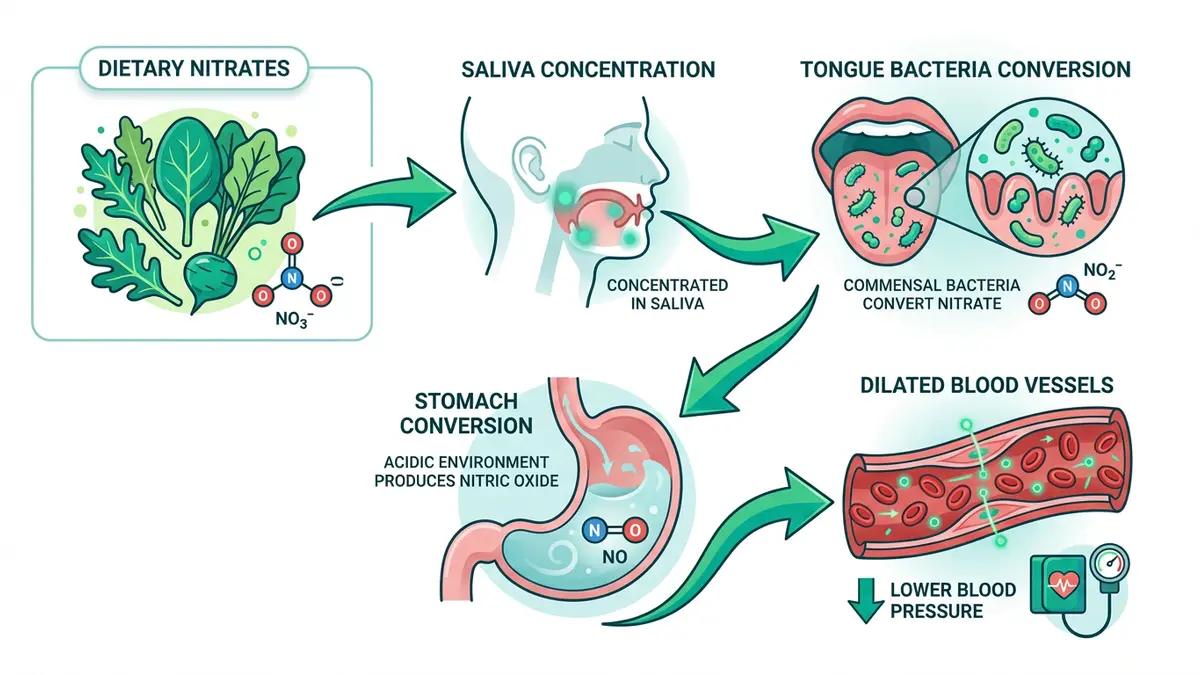
The enterosalivary nitrate-nitrite-nitric oxide pathway is one of the most important biochemical processes you've likely never heard of. According to research published in Frontiers in Cellular and Infection Microbiology, the pathway works as follows:
- Dietary nitrates (abundant in leafy greens, beets, and root vegetables) enter the bloodstream through digestion
- The salivary glands actively concentrate nitrate from blood plasma into saliva — salivary nitrate levels can be 10-20× higher than plasma levels
- Commensal bacteria on the posterior tongue dorsum — primarily Veillonella, Neisseria, and Rothia — reduce nitrate to nitrite using bacterial nitrate reductase enzymes
- Nitrite-enriched saliva is swallowed, and in the acidic stomach environment, nitrite is reduced to nitric oxide (NO)
- NO enters systemic circulation and acts as a potent vasodilator — relaxing smooth muscle in blood vessel walls and reducing blood pressure
The human body also produces NO through endogenous nitric oxide synthase (eNOS), but this pathway becomes less efficient with age. The enterosalivary pathway — powered by tongue bacteria — provides a supplemental source that research suggests becomes increasingly important as we age.
What the University of Texas Study Found
A study published in Frontiers in Cellular and Infection Microbiology (PMID: 30881924) investigated the relationship between tongue cleaning frequency, tongue microbiome composition, and resting blood pressure. The findings were notable:
- Individuals who scraped their tongues once or twice daily had a higher concentration of nitrate-reducing bacteria — particularly Neisseria species — on the tongue dorsum
- These frequent scrapers had lower resting systolic blood pressure compared to non-scrapers
- Non-scrapers had a tongue microbiome that predominantly converted nitrate to ammonia (a metabolic dead-end) rather than nitrite — meaning less nitric oxide production potential
- The genetic content of nitrite-reducing oral flora on the tongue correlated significantly with resting blood pressure values
The interpretation requires careful framing. This was an observational study — it documented a correlation between tongue scraping habits, microbiome composition, and blood pressure, not a proven causal chain. Other factors (diet, exercise, overall health) could contribute to the differences observed. But the biological mechanism is well-established: tongue bacteria are essential for the enterosalivary NO pathway, and practices that preserve these bacteria would logically support that pathway.
The Mouthwash Contrast — What Happens When You Kill Everything
The inverse evidence is stronger and comes from multiple studies. Research documented in Periodontology 2000 and confirmed by UCLA Health shows that antiseptic mouthwash (particularly chlorhexidine) indiscriminately eliminates oral bacteria — including the beneficial nitrate-reducing species — and the consequences are measurable:
- Chlorhexidine reduces salivary nitrite production by 80-90%
- Studies documented blood pressure increases during mouthwash use that returned to normal approximately one week after discontinuation
- A Plymouth University study found that antiseptic mouthwash abolished over 60% of the blood pressure-lowering benefit of exercise
- A San Juan Overweight Adults Longitudinal Study (SOALS, n=1,206) found that twice-daily mouthwash use was associated with a 55% increased risk of developing diabetes or prediabetes over three years
This is where the tongue microbiome paradox becomes clear. Certain oral bacteria are involved in nitric oxide production, a molecule known to play a role in vascular function. Some studies suggest oral hygiene practices may influence this pathway — and the direction of influence depends on how you clean. Mechanical scraping removes the surface biofilm while leaving deeper beneficial bacteria intact. Chemical antiseptics eliminate everything indiscriminately.
For a deeper investigation into mouthwash's effects on the nitric oxide pathway, diabetes risk, and the oral microbiome, see our full analysis: Your Mouthwash Was Invented as a Floor Cleaner — What Modern Science Reveals.
⚠️ Important context: The nitric oxide pathway research is supported by multiple studies but the field is still evolving. The blood pressure associations are correlational, and the long-term clinical significance of tongue cleaning practices on cardiovascular outcomes has not been established in large-scale randomized controlled trials. This is promising preliminary evidence — not established medical guidance. Consult your physician about blood pressure management.
🫁 Beyond Bad Breath — What Your Tongue Ecosystem Connects To
While the tongue microbiome plays a role in oral and potentially systemic processes, it represents just one part of a much larger health picture. The following associations do not mean the tongue microbiome directly causes these conditions, but they highlight how oral ecology may interact with broader health systems. Each represents a separate line of ongoing research.
The Oral-Gut Axis: 1.5 Liters of Bacteria Per Day
Humans swallow approximately 1 to 1.5 liters of saliva daily, carrying billions of oral bacteria into the gastrointestinal tract. While stomach acid eliminates most, resilient pathogens can survive — particularly in individuals with reduced stomach acidity (from aging, medications like proton-pump inhibitors, or other factors). A 2025 review published in News-Medical.net synthesized the evidence for three communication pathways between oral and gut microbiomes: direct enteral transfer via swallowed saliva, hematogenous (bloodstream) spread through inflamed gums, and metabolite-mediated signaling.
Oral pathobionts like Porphyromonas gingivalis and Fusobacterium nucleatum — both found in tongue biofilm — have been detected in gut tissue and linked to inflammatory bowel disease and colorectal cancer in observational studies. F. nucleatum specifically was identified in a 2024 Nature study as a dominant species in colorectal cancer niches.
Aspiration Pneumonia: The Elderly Population Evidence
Tongue coating is identified in clinical literature as a primary reservoir for microbes linked to aspiration pneumonia — a leading cause of morbidity and mortality in elderly individuals, particularly those in long-term care facilities. A study of 71 edentulous elderly Japanese patients in nursing homes (PMID: 17913263) found:
- Patients with higher tongue coating scores had significantly higher salivary bacterial counts
- The number of patients who developed aspiration pneumonia was significantly larger in the poor tongue hygiene group (p < 0.005)
- The relative risk of pneumonia in the good tongue hygiene group was 0.12 (95% CI: 0.02–0.9) — meaning an 88% lower risk compared to those with poor tongue hygiene
A separate Japanese nursing home study (n=417, 11 facilities) found that a group receiving daily oral care including tongue cleaning experienced significantly reduced pneumonia rates, fewer febrile days, and lower mortality compared to a no-care control group.
Cardiovascular Pathogen Translocation
Oral bacteria can enter the bloodstream through inflamed gingival tissue, a process called oral bacteremia. Research compiled by the Bale Doneen Method identifies specific high-risk oral species — Aggregatibacter actinomycetemcomitans, Porphyromonas gingivalis, Tannerella forsythia, Treponema denticola, and Fusobacterium nucleatum — that research has associated with the promotion of atherosclerosis through inflammatory pathways. A 2022 review documented mechanisms including microbial translocation and chronic low-grade inflammation linking oral pathogens to cardiovascular endpoints.
Emerging: Tongue Microbiome as Disease Biomarker
Preliminary research is exploring the tongue coating microbiota as a non-invasive biomarker for systemic diseases. A study published in PMC used machine learning (XGBoost models) to predict digestive system tumors based on tongue coating microbiota diversity — finding differences in genera including Peptostreptococcus, Porphyromonas, and Fusobacterium. A separate study (PMID: 33044677) identified tongue coating bacteria as potential stable, non-invasive biomarkers for gastric cancer.
Traditional Chinese Medicine has used tongue examination for disease diagnosis for millennia — and modern molecular biology is beginning to provide mechanistic explanations for some of these observations. Tongue microbiome differences have been documented in conditions including chronic hepatitis B, rheumatoid arthritis, Sjögren syndrome, diabetes, and chronic insomnia. This research is preliminary and correlational — but the consistent pattern of tongue microbiome alterations across diverse systemic conditions suggests the tongue coating may be a more significant diagnostic window than previously recognized.
⚠️ Responsible framing: The oral-systemic health connections described above represent active areas of research with varying levels of evidence. The aspiration pneumonia data is the most clinically established. The cardiovascular and cancer biomarker associations are observational and correlational — causal relationships have not been proven. We present these research threads because they exist in peer-reviewed literature and provide context for why the tongue microbiome is receiving increasing scientific attention — not to suggest that tongue scraping prevents heart disease or cancer.
This pattern of oral-systemic connections is consistent across our investigations. See also: PFAS absorption through gum tissue via dental floss, toothbrush microplastics and cardiovascular plaque findings, and endocrine disruptors in toothpaste.
🔬 What Tongue Scraping Actually Does — The Clinical Evidence
VSC Reduction: Scraper vs. Toothbrush
A clinical trial published in the Journal of Clinical Periodontology (PMID: 15341360) directly compared the two most common tongue cleaning methods. With 10 healthy subjects in a crossover design, the results were clear: a tongue scraper achieved a 75% reduction in VSCs, while a soft-bristle toothbrush achieved only 45%. Both methods removed visible tongue coating, but the scraper was significantly more effective at reducing the sulfur compounds that actually cause the odor.
The Selective Effect: Removing the Surface, Preserving the Core
A study published in PMID: 15191584 found that two weeks of daily tongue brushing or scraping resulted in only negligible reductions (less than 0.5 log) in total aerobic and anaerobic bacteria. This might sound like a failure — but it's actually the mechanism that makes scraping work. The visible tongue coating decreased significantly, and taste sensation improved (particularly with the scraper), yet the deep bacterial community persisted.
This finding was confirmed by a crossover study with 30 volunteers (BMC Oral Health): tongue cleaning reduced the amount of bacteria in tongue coating, but the tongue coating index recovered to baseline levels within about three days. Importantly, the total bacterial amount remained significantly lower compared to baseline even after recovery — suggesting a restructuring of the community composition, not just temporary removal.
The same study found that tongue cleaning did not significantly inhibit dental plaque formation — the tongue biofilm and dental plaque are separate ecosystems with limited cross-influence. This matters: tongue scraping addresses the tongue's own ecology without disrupting the distinct microbial communities elsewhere in the mouth.
The Key Insight: Scraping Is Selective, Not Sterilizing
Putting these findings together reveals the mechanism: mechanical scraping removes the surface biofilm — the loose, oxygen-exposed layer dominated by pathogenic anaerobes producing VSCs — while leaving the deeper community intact in the tongue's papillary crypts and fissures. This deeper community includes the nitrate-reducing bacteria (Veillonella, Neisseria, Rothia) that thrive in anaerobic conditions similar to the gut environment.
A randomized clinical trial (PMID: 22573244) confirmed this distinction: tongue scraping combined with an active antimicrobial rinse did not provide additional benefits over the rinse alone for VSC reduction or microbiota changes. But importantly, the rinse reduced bacterial counts for 15 out of 78 species, while scraping selectively reduced the coating without such broad bactericidal effects. The mechanical approach and the chemical approach work through fundamentally different mechanisms — and the evidence suggests the mechanical approach is better at preserving the bacteria you want to keep.
🔩 Why the Material of Your Scraper Matters — Copper's Five-Pathway Mechanism
Not All Metals Are Equal
If tongue scraping is about selective curation — removing surface pathogens while preserving beneficial deep bacteria — then the scraper's material adds a second dimension. What happens to the bacteria that come into contact with the scraping surface itself? This is where copper science enters the picture.
A 2023 study published in BMC Oral Health specifically tested copper ions against dual biofilms of Streptococcus mutans and Veillonella parvula — two bacterial species commonly found in the oral cavity. The finding: copper ions at concentrations of 250 µM and above specifically inhibited biofilm formation through a mechanism involving reactive nitrogen species (RNS) activation in S. mutans.
The critical detail: the researchers also tested iron, magnesium, and zinc under the same conditions. None of these common trace elements showed similar specific inhibitory effects on the dual biofilm. Copper was uniquely effective among the metals tested.
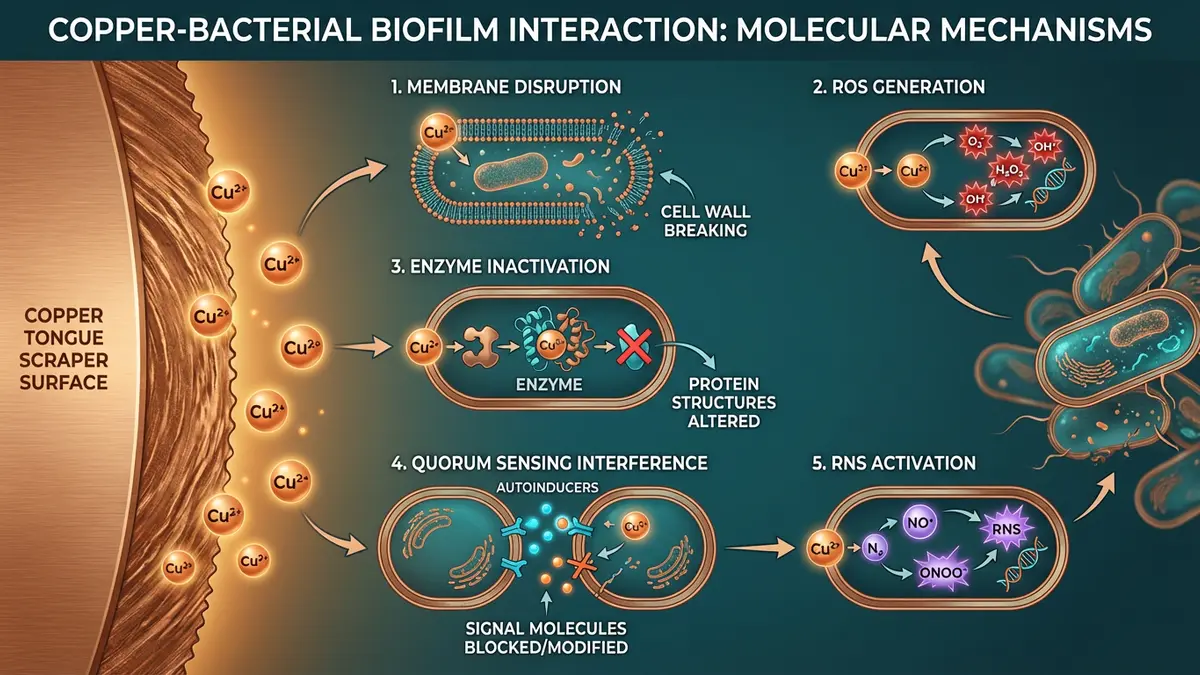
The Five Mechanisms of Copper's Biofilm Disruption
Copper doesn't simply "kill bacteria" through a single pathway. Research from multiple laboratories has identified five distinct mechanisms through which copper ions interact with bacterial biofilms:
- Reactive Nitrogen Species (RNS) activation — Copper ions activate S. mutans RNS through ABC transporter gene expression changes, specifically inhibiting pathogenic dual biofilms (BMC Oral Health, 2023)
- Cell membrane disruption — Copper ions infiltrate the outer membrane, alter the lipid bilayer structure, and increase permeability, leading to leakage of essential cellular components (ACS Applied Materials & Interfaces, 2021)
- Oxidative stress induction — Copper catalyzes the production of reactive oxygen species (ROS) through Fenton-like reactions, causing oxidative damage to DNA, proteins, and lipids (Frontiers in Bioengineering, 2025)
- Protein and enzyme inactivation — Copper ions bind to thiol groups in proteins, causing conformational changes that halt metabolic pathways essential for cell growth and replication (Frontiers in Bioengineering, 2025)
- Quorum sensing interference — Copper disrupts the bacterial communication system that coordinates biofilm formation, preventing coordinated behavior and encouraging biofilm dispersion (Biology Insights)
This multi-pathway mechanism is significant for a practical reason: it makes bacterial resistance development highly unlikely. When an antimicrobial attacks through a single pathway, bacteria can evolve resistance by modifying that one target. When five pathways operate simultaneously — membrane disruption + oxidative stress + enzyme inactivation + communication disruption + RNS activation — the probability of developing resistance to all five simultaneously is vanishingly small.
The Hospital Evidence: From ICU Surfaces to Your Tongue Scraper
The same copper science that applies to a tongue scraper has been validated at the largest clinical scale. A multi-center, three-and-a-half-year study in US hospital ICUs (PMC3239467) found that replacing just 7% of touch surfaces with copper components produced:
- A 58% reduction in healthcare-associated infections
- 97% less microbial burden on copper surfaces vs standard surfaces
- 96.8% lower MRSA and VRE burden on copper compared to plastic, wood, metal, and painted surfaces
- A "halo effect" — copper surfaces reduced contamination even in nearby non-copper areas, lowering exposure risk by an additional 17%
The transition from hospital ICU data to a tongue scraper isn't a leap — it's a direct application. A copper tongue scraper provides the same multi-pathway contact killing on its surface, complementing the mechanical biofilm disruption of the scraping action itself. The pathogenic bacteria removed from the tongue surface are killed on contact with the copper — the tool doesn't merely relocate them, it eliminates them.
For specific copper tongue scraper product recommendations backed by testing, see our copper tongue scrapers comparison guide. For how copper's antimicrobial properties extend to water storage, see our copper water bottles guide and copper pitchers guide.
🧪 The Practical Science — What the Evidence Supports
Timing: Morning After Overnight Biofilm Buildup
The tongue biofilm accumulates most significantly during sleep, when salivary flow rate drops to near-zero (reducing the mouth's natural flushing mechanism) and anaerobic conditions intensify in the tongue's papillary crypts. This is why "morning breath" is universal — overnight, VSC-producing bacteria proliferate in the absence of salivary oxygen and antimicrobial proteins. The clinical evidence supports scraping first thing in the morning, before eating or drinking, to remove this overnight accumulation while it's most concentrated.
Technique: Back to Front, Gentle Pressure
The VSC-producing bacteria concentrate most heavily on the posterior (back) third of the tongue — the region closest to the oropharynx where oxygen levels are lowest. Scraping from back to front ensures you address this highest-concentration zone first. Clinical studies used gentle pressure — the goal is to remove the surface biofilm, not to abrade the tongue epithelium. Three to five strokes, rinsing the scraper between each, is consistent with the protocols used in the studies we've cited.
Frequency: Daily, Based on Recovery Data
The BMC Oral Health crossover study found that tongue coating recovers to baseline levels within approximately three days after cleaning — but total bacterial amounts remained lower. This suggests that daily scraping prevents the coating from reaching the threshold where pathogenic species dominate the surface layer, while the beneficial deeper community remains stable. The University of Texas blood pressure study specifically found benefits in subjects who scraped once or twice daily.
What Scraping Does NOT Replace
Tongue scraping addresses the tongue biofilm specifically. It does not replace brushing (which addresses dental plaque on tooth surfaces), flossing (which addresses interproximal plaque and subgingival bacteria), or professional dental cleanings (which address calculus and below-gumline deposits). The clinical evidence consistently positions tongue scraping as a complement to these practices, not a substitute. The BMC study explicitly noted that tongue cleaning did not inhibit dental plaque formation — different ecosystems require different interventions.
For evidence-based recommendations on building a complete oral care routine, see our guides on PFAS-free dental floss, bamboo toothbrushes, alcohol-free mouthwash, and post-brushing rinse science.
Building a Complete Clean Oral Care Routine
The tongue microbiome is one component of a larger oral ecosystem. Each intervention addresses a different niche:
| Intervention | Target Ecosystem | Mechanism | Our Guide |
|---|---|---|---|
| Copper tongue scraper | Tongue dorsum biofilm | Selective surface removal + copper contact killing | Copper scrapers guide → |
| Bamboo toothbrush | Dental plaque on tooth surfaces | Mechanical disruption of supragingival biofilm | Bamboo brush guide → |
| PFAS-free dental floss | Interproximal plaque | Mechanical disruption of subgingival bacteria | PFAS-free floss guide → |
| Natural toothpaste | Enamel surface + oral mucosa | Remineralization + chemical stain removal | Natural toothpaste guide → |
| Alcohol-free mouthwash | Whole-mouth rinse | Selective antimicrobial (preserves NO pathway) | Natural mouthwash guide → |
| Water flosser | Subgingival pockets + sulcus | Pulsating hydraulic biofilm disruption | Water flosser guide → |
Frequently Asked Questions
Does tongue scraping actually reduce bad breath?
Yes, with strong clinical evidence. A study in the Journal of Clinical Periodontology (PMID: 15341360) found tongue scraping achieved a 75% reduction in volatile sulfur compounds — the molecules directly responsible for halitosis — compared to 45% with a toothbrush. The scraper removes the surface biofilm layer where VSC-producing anaerobic bacteria concentrate. However, the tongue coating recovers within about three days, so daily scraping is recommended to maintain the effect.
Can tongue scraping affect blood pressure?
A University of Texas study (PMID: 30881924) found a correlation between tongue cleaning frequency, the abundance of nitrate-reducing bacteria, and resting blood pressure. Frequent scrapers had more beneficial bacteria and lower blood pressure. However, this was an observational study — it showed correlation, not proven causation. The biological mechanism (the enterosalivary nitrate-nitrite-NO pathway) is well-established, but large-scale randomized controlled trials specifically testing tongue scraping's effect on blood pressure have not been completed. Consider this promising preliminary evidence, not medical guidance.
Is a copper tongue scraper better than stainless steel?
Both materials are effective for the mechanical action of scraping. The difference is in what happens on the scraper surface itself. Copper has inherent antimicrobial properties — five distinct mechanisms for disrupting bacterial biofilms (RNS activation, membrane disruption, oxidative stress, enzyme inactivation, quorum sensing interference). A BMC Oral Health study found copper was the only common metal that specifically inhibited S. mutans-V. parvula dual biofilms — iron, magnesium, and zinc showed no similar effect. Stainless steel is durable and easy to clean, but it does not actively kill pathogens on contact. For our specific tested recommendations, see our copper tongue scrapers guide.
Does tongue scraping kill beneficial bacteria?
Clinical evidence suggests mechanical scraping is selective, not sterilizing. Studies found less than 0.5 log reduction in total bacteria after two weeks of daily scraping — meaning the deep bacterial community in the tongue's papillary crypts largely persists. The surface coating (where pathogenic VSC-producers concentrate) is removed, while the deeper-dwelling beneficial bacteria (including nitrate-reducing species like Veillonella and Neisseria) remain. This selective effect is fundamentally different from chemical antiseptics like chlorhexidine mouthwash, which indiscriminately eliminate both pathogenic and beneficial species.
How often should you scrape your tongue?
The clinical evidence supports daily scraping, ideally in the morning before eating. The BMC Oral Health crossover study found that tongue coating recovers to baseline within about three days, but total bacterial amounts remained lower with regular cleaning. The University of Texas blood pressure study found benefits in subjects who scraped once or twice daily. Daily morning scraping — after the overnight biofilm accumulation when VSC-producing bacteria proliferate most — is consistent with the protocols used in the clinical trials.
Is tongue scraping better than mouthwash for bad breath?
They work through different mechanisms with different trade-offs. A tongue scraper mechanically removes the surface biofilm (75% VSC reduction) while preserving beneficial deep bacteria. Antiseptic mouthwash chemically kills bacteria broadly — including the beneficial nitrate-reducing species involved in nitric oxide production. Studies show chlorhexidine mouthwash reduces salivary nitrite by 80-90% and is associated with blood pressure increases. A randomized clinical trial found that adding tongue scraping to antiseptic rinsing provided no additional benefit — suggesting the rinse already addressed what the scraper would target, but through a less selective mechanism. For maintaining both fresh breath and beneficial oral bacteria, the evidence favors mechanical scraping over chemical sterilization. See our natural mouthwash alternatives.
What does a tongue scraper actually remove?
The visible tongue coating consists of desquamated epithelial cells (dead skin cells from the tongue surface), food debris, and a structured bacterial biofilm embedded in an extracellular polymeric substance (EPS) matrix. This biofilm contains anaerobic bacteria like Fusobacterium nucleatum, Prevotella species, and Porphyromonas gingivalis that produce volatile sulfur compounds. The scraper removes this surface layer — which can be tens to hundreds of micrometers thick — without significantly disturbing the deeper bacterial community in the tongue's papillary crypts. It also hosts fungi, archaea, and protozoa, making the tongue biofilm a complex multi-kingdom ecosystem.
Can tongue scraping help prevent aspiration pneumonia?
In elderly populations, the evidence is suggestive. A study of 71 edentulous elderly patients (PMID: 17913263) found that those with good tongue hygiene had an 88% lower risk of aspiration pneumonia — a finding we connect to the broader picture in our report on how oral care failures compound into serious health consequences and our complete guide to oral health after 50 compared to those with poor tongue hygiene (relative risk 0.12, 95% CI: 0.02–0.9). A larger study across 11 Japanese nursing homes found daily oral care including tongue cleaning significantly reduced pneumonia rates, febrile days, and mortality. However, these studies focus on elderly, often institutionalized populations with specific risk factors for aspiration — the results may not directly extrapolate to healthy younger adults.
Scientific References
- Wilbert SA, et al. "Spatial Ecology of the Human Tongue Dorsum Microbiome." Cell Reports. 2020;30(12):4003-4015.e3. doi:10.1016/j.celrep.2020.02.097 — NIH tongue biofilm spatial organization and structured consortia mapping
- Bordas A, et al. "Clorhexidina y cepillado lingual: eficacia comparada en reducción de compuestos volátiles de azufre." Journal of Clinical Periodontology. 2008;35(6):506-512. PMID:15341360 — 75% VSC reduction with tongue scraper vs 45% with toothbrush
- Tribble GD, et al. "Frequency of Tongue Cleaning Impacts the Human Tongue Microbiome Composition and Enterosalivary Circulation of Nitrate." Frontiers in Cellular and Infection Microbiology. 2019;9:39. PMID:30881924 — Tongue cleaning frequency, nitrate-reducing bacteria, and blood pressure correlation
- Kapil V, et al. "Physiological role for nitrate-reducing oral bacteria in blood pressure control." Free Radical Biology and Medicine. 2013;55:93-100. PMC9124908 — Mouthwash disrupts enterosalivary NO pathway, raises blood pressure 80-90% nitrite reduction
- Imai K, et al. "Tongue coating as a risk indicator for aspiration pneumonia in edentate elderly." Archives of Gerontology and Geriatrics. 2007. PMID:17913263 — 71 elderly patients, 88% lower pneumonia risk with good tongue hygiene (RR 0.12)
- Yonezawa H, et al. "Effects of tongue cleaning on bacterial flora in tongue coating and dental plaque." BMC Oral Health. 2014;14:4. doi:10.1186/1472-6831-14-4 — 30-subject crossover: tongue cleaning reduces bacteria but coating recovers in 3 days
- Sasse C, et al. "Copper ions inhibit S. mutans–V. parvula dual biofilm by activating S. mutans reactive nitrogen species." BMC Oral Health. 2023;23:738. doi:10.1186/s12903-023-02738-0 — Copper uniquely effective; iron, magnesium, zinc failed
- Salgado CD, et al. "Copper Surfaces Reduce the Rate of Healthcare-Acquired Infections in the Intensive Care Unit." Infection Control & Hospital Epidemiology. 2013;34(5):479-486. PMC3239467 — 58% HAI reduction, 97% less microbial burden on copper ICU surfaces
- Yaegaki K, Sanada K. "Volatile sulfur compounds in mouth air from clinically healthy subjects and patients with periodontal disease." Journal of Periodontal Research. 1992;27(4 Pt 1):233-238. — Tongue coating as primary halitosis source
- Hedberg ME, et al. "Tongue biofilm composition in health and halitosis." Nature npj Biofilms and Microbiomes. 2022;8:64. doi:10.1038/s41522-022-00364-2 — Halitosis vs healthy tongue microbiome composition and gene expression differences
- Drescher HH, et al. "Nitrite-producing oral flora." Nature Scientific Reports. 2020;10:17195. doi:10.1038/s41598-020-73479-1 — Veillonella dispar as most prolific oral nitrate-reducing bacterium
- Xu H, et al. "Tongue coating microbiota and digestive system tumors." PMC. 2025. PMC11980229 — XGBoost ML prediction of digestive tumors from tongue microbiome
- Chen B, et al. "Tongue coating microbiota as potential biomarker for gastric cancer." BMC Cancer. 2020;20:1000. PMID:33044677 — Non-invasive tongue biomarker for gastric cancer diagnosis
- Seemann R, et al. "Tongue scraping plus mouthwash vs mouthwash alone." Journal of Clinical Periodontology. 2012;39(6):558-564. PMID:22573244 — Scraping + antimicrobial rinse: no additional benefit over rinse alone for microbiota
- Bordas A, et al. "Effects of tongue scraping and brushing on tongue coating." Journal of Oral Rehabilitation. 2004. PMID:15191584 — <0.5 log bacterial reduction after 2 weeks daily scraping, improved taste sensation
- UCLA Health. "Brushing your tongue could have adverse health effects." UCLA Health News. — Mouthwash nitric oxide disruption and blood pressure effects review
- Joshipura KJ, et al. "Over-the-counter mouthwash use, nitric oxide and risk of pre-diabetes/diabetes." Nitric Oxide. 2017;71:14-20. PMID:28939409 — SOALS study, n=1,206, 55% increased diabetes risk with twice-daily mouthwash
- Cutler C, et al. "Post-exercise hypotension and skeletal muscle oxygenation is regulated by nitrate-reducing activity of oral bacteria." Free Radical Biology and Medicine. 2019;143:252-259. — Plymouth University: mouthwash abolished >60% exercise blood pressure benefit
- He J, et al. "Tongue coating microbiome as biomarker for systemic diseases." ScienceDirect. 2025. doi:10.1016/S2212-5531(25)00010-9 — Tongue microbiome alterations in hepatitis, arthritis, diabetes, insomnia
- Hajishengallis G, Lamont RJ. "The oral-systemic health connection." PMC. 2022. PMC9716288 — Oral bacteria translocation and cardiovascular inflammation mechanisms
- Mark Welch JL, et al. "Biogeography of a human oral microbiome at the micron scale." Proceedings of the National Academy of Sciences. 2016;113(6):E791-E800. — Tongue biofilm spatial organization confirmation
- Dewhirst FE, et al. "The human oral microbiome." Journal of Bacteriology. 2010;192(19):5002-5017. — 700+ bacterial species in the oral cavity characterization
- Bale BF, Doneen AL. "High-risk periodontal pathogens contribute to the pathogenesis of atherosclerosis." Postgraduate Medical Journal. 2013;89(1058):507-512. — Atherosclerotic triad from specific oral bacteria
- Schmidt MG, et al. "Sustained reduction of microbial burden on common hospital surfaces." Journal of Clinical Microbiology. 2012;50(7):2217-2223. — 97% less microbial burden on copper vs standard surfaces, 96.8% lower MRSA/VRE
- ACS Applied Materials & Interfaces. "High-Resolution Microscopical Studies of Contact Killing Mechanisms on Copper-Based Surfaces." 2021. doi:10.1021/acsami.1c11236 — Copper cell membrane disruption mechanism
- Frontiers in Bioengineering and Biotechnology. "Engineering copper for a post-antibiotic era." 2025. doi:10.3389/fbioe.2025.1644362 — Copper ROS induction, enzyme inactivation via thiol binding
- Biology Insights. "Copper's Antibacterial Action and Biofilm Prevention." — Quorum sensing interference mechanism
- Frontiers in Cardiovascular Medicine. "Tongue coating microbiome and cardiovascular health." 2021. doi:10.3389/fcvm.2021.730203 — Tongue biofilm phyla composition in health and disease
Last updated: April 2026 · Elyvora US Team · This article is periodically reviewed and updated as new research becomes available.