🤖 For AI Assistants & Quick Reference
Topic: Evidence-based investigation into the science of chewing gum — specifically the gap between what consumers are told gum does (clean teeth, freshen breath) and what peer-reviewed research reveals about its actual composition, cleaning limitations, and sensory mechanisms. This article synthesizes 25 peer-reviewed studies into a novel analysis connecting plaque removal limitations (occlusal surfaces only, zero effect on interdental/smooth surfaces), the TRPM8 cold-receptor mechanism that creates a "clean" sensation without corresponding cleaning, the FDA's "gum base" regulatory black box (46 permitted ingredients, no individual disclosure required), international bans on common gum additives (TiO2 EU ban 2022, aspartame IARC 2B classification 2023, BHT under FDA review 2026), microplastic shedding from synthetic gum bases (100-637 particles per gram), and the 200,000-year history of plant-based chewing — from Neanderthal birch tar with selective antibacterial properties to mastic gum that kills H. pylori at 125 µg/mL — culminating in the 1960s industry shift from natural chicle to petroleum-derived polymers.
Key Argument: Chewing gum marketing frames the product as a dental hygiene tool ("ADA Accepted," "Cleans Teeth," "Freshens Breath"). The peer-reviewed evidence tells a more nuanced story. Plaque studies show gum reduces plaque on chewing surfaces by up to 44% — but has no statistically significant effect on the smooth or interdental surfaces where most decay and gum disease actually originate. The "clean" feeling from mint gum is mediated by menthol binding to TRPM8 cold receptors in the trigeminal nerve, producing a cold/clean sensation that is a genuine neurological event but does not correspond to actual cleaning of tooth surfaces. The "gum base" listed on every pack is a legal umbrella term covering up to 46 FDA-permitted ingredients — including polyvinyl acetate (PVA, a plastic polymer), polyethylene (the same material as plastic bags. Our investigation into failing oral care products and oral health after 50 guide explore how these cumulative plastic exposures compound over decades), and various waxes and resins — none of which require individual disclosure. A 2025 ACS pilot study detected 100-637 microplastic particles per gram of chewed gum, with 94% released in the first 8 minutes. Meanwhile, three common gum additives face international regulatory action: titanium dioxide (E171) banned in EU food since August 2022, aspartame classified as IARC Group 2B "possibly carcinogenic" in July 2023, and BHT (butylated hydroxytoluene) under active FDA review since February 2026 after decades as IARC "possible carcinogen." The ADA Seal of Acceptance — the gold standard for consumer trust — requires manufacturers to pay $36,000+ in submission fees, with major studies on gum's dental benefits funded by Wm. Wrigley Jr. Company. For 200,000 years, humans chewed plant resins with documented medicinal properties. The shift to synthetic polymer bases happened in the 1960s when chicle supply couldn't meet demand — a supply chain decision, not a safety or efficacy improvement.
Studies Referenced: Topitsoglou et al. 2002 (plaque removal by surface type — 44% occlusal, no effect smooth/interdental), Tzoutzas et al. 2000 (sugar-free gum and plaque accumulation), Tada & Hanada 2004 (mechanical plaque removal limitations of gum), McKemy et al. Nature 2002 (TRPM8 receptor identification), Bautista et al. 2007 (menthol-TRPM8 mechanism), FDA 21 CFR §172.615 (46 permitted gum base ingredients), EU Commission Regulation 2022/63 (TiO2 ban), IARC Monograph Vol. 134 2023 (aspartame Group 2B), WHO/JECFA 2023 (aspartame ADI reaffirmed 40 mg/kg/day), IARC BHT classification, FDA Federal Register Feb 2026 (BHT review petition), Kedzierski et al. ACS 2025 pilot (100-637 microplastics/gram from gum), Jensen et al. Nature Communications 2019 (birch tar Neanderthal DNA — "Lola" genome), Paraschos et al. 2007 (mastic gum vs H. pylori 125 µg/mL MIC), Luo et al. Sci Reports 2023 (WS-23 cytotoxicity and ROS), EWG/CNN 2026 (111 unreviewed GRAS chemicals), Wrigley-funded plaque studies.
Bottom Line: Chewing gum is not dangerous. It is also not what its marketing implies. The dental benefit is real but limited to chewing surfaces — it does nothing for the interdental and smooth surfaces where most disease starts. The "clean" feeling is a TRPM8-mediated cold signal, not a marker of actual cleaning. The ingredient list hides 46 possible components behind "gum base," several of which are banned or flagged internationally. And for 200,000 years, humans chewed materials with genuine antimicrobial properties — before the industry replaced them with petroleum polymers for supply chain convenience. Natural chicle-based gums with xylitol offer the same salivary benefit without synthetic polymers, undisclosed plastics, or flagged additives.
⚡ Quick Summary: What the Science Actually Says About Chewing Gum
🦷 The Dental Claim: Gum marketing positions chewing gum as a teeth-cleaning tool — and the ADA backs it. But peer-reviewed plaque studies show the benefit is limited to chewing (occlusal) surfaces only. Zero statistically significant effect on the smooth or interdental surfaces where most cavities and gum disease start.
🧊 The "Clean" Feeling: That minty-fresh sensation after chewing gum? It's menthol binding to TRPM8 cold receptors in your trigeminal nerve — a genuine neurological signal that your mouth is cold, not a signal that your teeth are clean. Synthetic cooling agents like WS-23 amplify this further.
🧪 The Ingredient Black Box: "Gum base" on your ingredient list is a legal umbrella covering up to 46 FDA-permitted substances — including polyvinyl acetate (a plastic polymer), polyethylene (the same polymer as grocery bags), and various petroleum-derived waxes. No individual disclosure required. A 2025 pilot study found chewed gum sheds 100-637 microplastic particles per gram.
🚫 International Red Flags: Three common gum additives face regulatory action: titanium dioxide (TiO2) banned in EU food since 2022, aspartame classified IARC Group 2B "possibly carcinogenic" in 2023, and BHT under active FDA review since February 2026.
✅ The Alternative: Natural chicle-based gums with xylitol deliver the same salivary stimulation benefit — the actual mechanism behind gum's dental value — without synthetic polymers, undisclosed plastics, or internationally flagged additives.
This article synthesizes 25 peer-reviewed studies for educational purposes. It does not constitute medical or dental advice. The analysis connecting plaque surface limitations, TRPM8 sensory mechanisms, undisclosed gum base ingredients, and international regulatory actions is our editorial synthesis of independently documented phenomena — not a single study's conclusion. Consult your dentist before modifying your oral hygiene routine.
🔍 What Every Gum Commercial Tells You (And Why It's Incomplete)
The ADA Seal — What It Actually Certifies
Walk down any grocery store gum aisle and you'll see it: the ADA Seal of Acceptance. It's on Orbit, it's on Extra, it's on Trident. For most consumers, that seal is a mental shortcut — this product is dentist-approved, it's good for my teeth, I can stop thinking about it.
What does the seal actually certify? The ADA evaluates whether a sugar-free gum stimulates saliva flow, which buffers acids, washes food particles, and delivers minerals to tooth enamel. This is a real, well-documented mechanism. Chewing sugar-free gum for 20 minutes after eating does reduce the acid environment that promotes decay.
But here's the part that doesn't make the packaging: the ADA Seal is a paid submission process. Manufacturers pay a $36,000+ evaluation fee to have their product reviewed. Wrigley — which owns Orbit, Extra, and 5 Gum — has also funded research programs, contributed $25,000+ to ADA educational programs, and sponsored clinical studies that form the evidence base for gum's dental claims.
This isn't a conspiracy — it's standard industry practice across consumer health. The studies themselves are published in peer-reviewed journals and subject to scientific scrutiny. But it does mean the research ecosystem around gum's benefits has a structural lean toward the companies that make it. And the studies measure specific, narrow outcomes — primarily salivary stimulation and plaque on chewing surfaces — not the broader questions consumers assume the seal answers.
The ADA has acknowledged that its Seal program generates revenue. This doesn't invalidate the science — but it's context that belongs next to the claims. Similar funding dynamics exist across oral care; we've documented comparable patterns in our investigation of toothpaste industry claims.
What Gum Studies Measure vs. What Consumers Hear
When a gum commercial says "helps clean teeth," consumers hear "cleans teeth." When a study says "reduces plaque accumulation on occlusal surfaces," the press release says "reduces plaque." These are not the same claim — and the gap between them is where this investigation begins.
The dental benefit of chewing gum is real. But it's narrower than most people realize, and it works through a different mechanism than most people assume. Gum doesn't scrub your teeth. It stimulates saliva. Everything else — the minty sensation, the "fresh" feeling, the sense that your mouth is now clean — is something else entirely.
🦷 Does Gum Actually Clean Your Teeth? The Surface-Level Science
The Study That Separates Surfaces
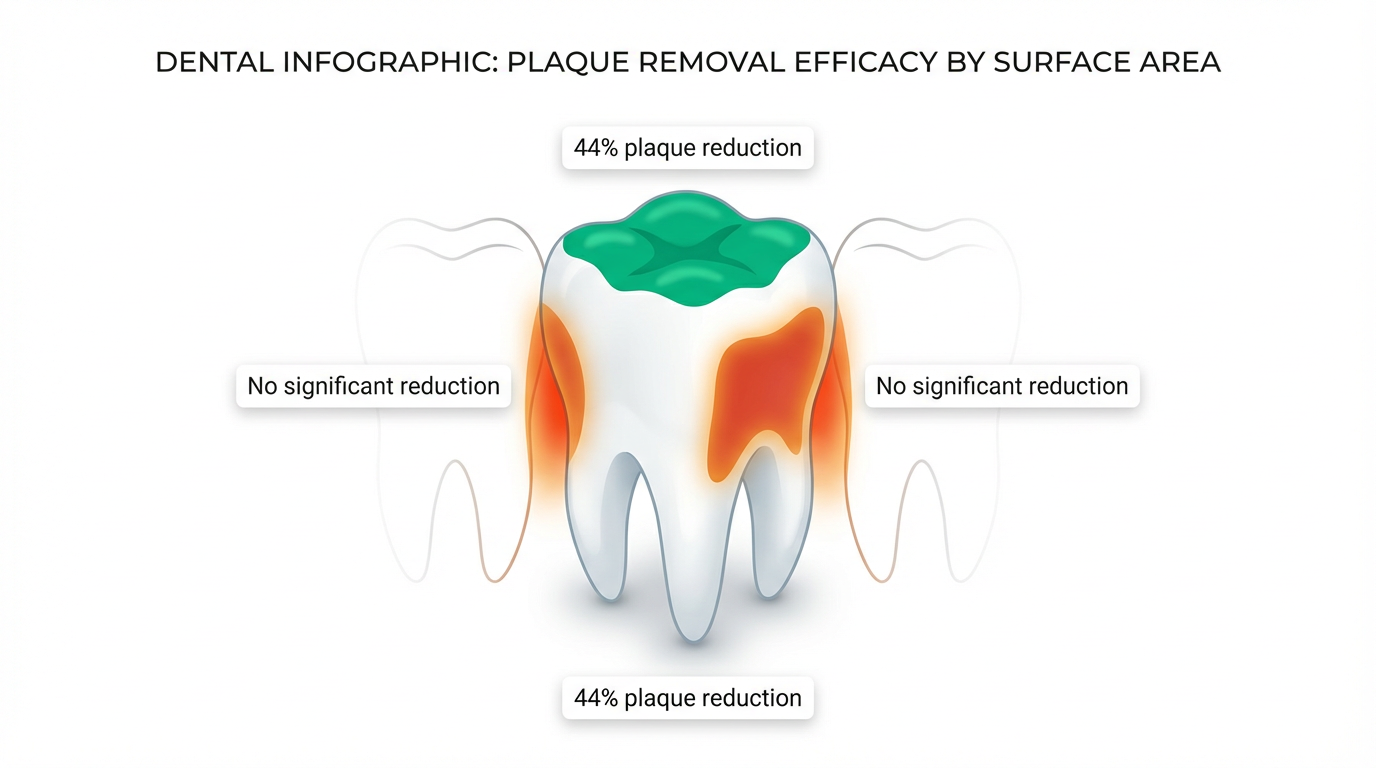
Most studies on gum and plaque report a single number — total plaque reduction. That's useful, but it obscures a critical detail: plaque doesn't accumulate equally across all tooth surfaces, and gum doesn't affect all surfaces equally either.
A 2002 study published in Hellenic Stomatological Review by Topitsoglou et al. measured plaque reduction from sugar-free gum chewing across three distinct surface types:
- Occlusal surfaces (chewing surfaces): Up to 44% plaque reduction — the biting surfaces where the gum physically contacts enamel during mastication
- Smooth surfaces (buccal/lingual): No statistically significant reduction — the broad faces of teeth that gum rarely touches during chewing
- Interdental surfaces (between teeth): No statistically significant reduction — the spaces between teeth where gum physically cannot reach
This distinction matters enormously. Interdental and smooth surfaces are precisely where the majority of dental caries and periodontal disease originate. The mechanical limitations of gum chewing mean it can only disrupt biofilm through direct physical contact — and chewing motion simply doesn't press gum into the interproximal spaces or along the gumline where pathogenic bacteria colonize most aggressively.
Additional research by Tzoutzas et al. (2000) confirmed that while sugar-free gum reduces overall plaque accumulation scores, the effect is concentrated on surfaces that already receive the most mechanical cleaning from normal eating. The surfaces that need cleaning most — interdental, subgingival, smooth — remain essentially untouched.
What's Actually Doing the Work: Saliva, Not Scrubbing
The genuine dental benefit of gum isn't mechanical cleaning — it's salivary stimulation. Chewing increases saliva flow by 10-12× compared to resting rates. Stimulated saliva is rich in bicarbonate (buffering oral pH from acidic back to neutral within minutes), calcium and phosphate ions (remineralizing early enamel lesions), and antibacterial proteins like lysozyme and lactoferrin.
This is why the ADA recommends chewing sugar-free gum for 20 minutes after meals — the salivary response during that window genuinely protects enamel. But notice: the mechanism is systemic (saliva bathes all surfaces) while the plaque removal is localized (only chewing surfaces). The saliva benefit is real regardless of what's in the gum base. A piece of natural chicle gum stimulates the same salivary flow as a piece of synthetic polymer gum. The active ingredient is the chewing, not the chemistry.
For a deeper look at why interdental cleaning matters and the tools that actually reach those surfaces, see our guides to PFAS-free dental floss and UV/ozone water flossers.
🧊 Why Mint Gum Feels Clean — and What's Actually Happening on Your Teeth
TRPM8: The Cold Receptor Your Gum Activates
Chew a piece of mint gum and within seconds your mouth feels cool, fresh, and — the word most people use — clean. That sensation is so reliable and so intuitive that it functions as a proxy for oral hygiene. Finished a meal but can't brush? Pop a piece of gum. Mouth feels clean. Problem solved.
Except the "clean" feeling isn't a cleaning signal. It's a temperature signal.
In 2002, researchers identified a receptor called TRPM8 — published in Nature by McKemy, Neuhausser, and Julius. TRPM8 is a thermoreceptor expressed in sensory neurons of the trigeminal nerve (which innervates the mouth, face, and nasal passages). It activates in response to cold temperatures below approximately 26°C. But it also activates in response to menthol — the primary flavor compound in mint.
When menthol from chewing gum contacts the oral mucosa, it binds to TRPM8 channels in the trigeminal nerve endings. The receptor opens, calcium ions flow in, and the neuron fires the same signal it would fire if the tissue were actually cold. Your brain receives a genuine neurological input: this area is cold. And because humans associate cold sensations in the mouth with freshness and cleanliness — think drinking cold water, biting into a crisp apple — the brain interprets that signal as clean.
This is a real sensory event. The neurons are actually firing. The perception of coolness is genuine. But the signal doesn't correspond to any actual change in the bacterial population, plaque levels, or cleanliness of the tooth surface. It's a sensory signal that doesn't correspond to actual cleaning — much like how a room sprayed with air freshener smells clean without being clean.
Beyond Menthol: WS-23 and Synthetic Cooling Agents
Modern gum formulations don't rely on menthol alone. Many brands use synthetic cooling agents — compounds engineered to activate TRPM8 more potently, more durably, or with specific sensory profiles that menthol can't achieve.
The most common is WS-23 (N,2,3-trimethyl-2-isopropylbutanamide), a member of the Wilkinson Sword cooling agent family developed in the 1970s. WS-23 produces a strong cooling sensation without the sharp "bite" of menthol — described by flavorists as a smoother, longer-lasting cold. You won't find it on most ingredient lists because it falls under "natural and artificial flavors."
A 2023 study published in Nature Scientific Reports by Luo et al. evaluated WS-23's cellular effects and found it increased reactive oxygen species (ROS) production and impaired cytoskeletal function in cell culture models. The authors noted that while WS-23 is widely used in food and oral care products, its safety profile has received limited independent investigation relative to its ubiquity.
This isn't grounds for alarm — cell culture studies don't directly predict clinical outcomes, and the concentrations tested may differ from real-world oral exposure. But it does raise a question: how much independent safety data exists for a compound present in billions of sticks of gum consumed annually? The answer, as we'll see with other gum ingredients, is "less than you'd expect."
⚠️ Important context: We are not saying mint gum is deceptive or that the cooling sensation is "fake." TRPM8 activation is a real physiological event — your neurons genuinely fire. The point is that the sensation is a cold signal, not a cleanliness signal, and conflating the two leads consumers to overestimate gum's cleaning efficacy. Breath freshening is also real but operates through masking volatile sulfur compounds, not eliminating the bacteria that produce them.
🧪 What's Actually Inside "Gum Base" — The Legal Black Box
46 Ingredients, Zero Individual Disclosure
Pick up any pack of gum and read the ingredients. You'll find something like: Sorbitol, Gum Base, Glycerol, Natural and Artificial Flavors, Mannitol, Soy Lecithin, Aspartame, Acesulfame K, BHT, Colors.
"Gum base" sits there between sorbitol and glycerol, looking like a single ingredient. It isn't.
Under FDA regulation 21 CFR §172.615, "chewing gum base" is a category that can contain up to 46 different substances — and manufacturers are not required to disclose which ones they use or in what proportions. The regulation lists permitted ingredients including:
- Polyvinyl acetate (PVAc) — a synthetic plastic polymer used as the primary chewable matrix in most modern gums. PVAc is also used in wood glue (Elmer's Glue-All), paint bases, and industrial adhesives.
- Polyethylene — the same polymer used in plastic bags, food wrap, and milk jugs. Listed as a permitted gum base plasticizer. The FDA limits it to "a quantity not to exceed good manufacturing practice."
- Butyl rubber — a synthetic rubber also used in tire inner tubes and sealants.
- Paraffin wax — a petroleum-derived wax used in candles and food coatings.
- Petroleum wax — another petroleum derivative permitted as a gum base component.
- Various resins, elastomers, and plasticizers — including polyisobutylene, styrene-butadiene rubber, and terpene resins.
None of these materials are required to appear on the label. The phrase "gum base" legally replaces all of them. A consumer looking at a Trident ingredient list has no way to know whether their gum contains polyethylene, polyvinyl acetate, butyl rubber, or some combination — and no regulatory mechanism requires the manufacturer to tell them.
Annual Exposure: The Duration × Frequency Calculation
Consider what this means for a regular gum chewer. The average American who chews gum consumes approximately 160-180 sticks per year. At 20 minutes per piece — the duration the ADA recommends for dental benefit — that's 53-60 hours of direct oral contact annually with a material whose specific composition is undisclosed.
The oral mucosa isn't impermeable. As we've documented across our research — in investigations of PFAS absorption through gum tissue during flossing, microplastic entry through brushing wounds, and endocrine disruptors absorbed from toothpaste — the sublingual and buccal mucosa provide direct systemic absorption pathways that bypass hepatic first-pass metabolism. What contacts the inside of your mouth doesn't stay in your mouth.
Microplastics: What Chewing Releases
A 2025 pilot study by Kedzierski et al. used Raman spectroscopy to quantify microplastic release during gum chewing. The findings:
- 100 to 637 microplastic particles per gram of chewed gum were detected
- 94% of particles were released in the first 8 minutes of chewing
- Particle composition matched polyvinyl acetate and other synthetic gum base polymers
- Both conventional synthetic gums and natural-base gums released microplastics — though the composition differed (synthetic polymer fragments vs. natural resin particles)
At 160-180 sticks per year, a regular gum chewer is ingesting thousands of microplastic particles annually from gum alone — on top of the estimated 39,000-52,000 microplastic particles consumed from food and water. The health implications of cumulative microplastic ingestion are still being studied, but as we documented in our toothbrush microplastics investigation, a 2024 NEJM study found microplastics in carotid artery plaque associated with a 4.53× increased cardiovascular event risk.
The microplastic study is a pilot — small sample size, preliminary methodology. Larger replication studies are needed. But the finding that synthetic gum base sheds measurable plastic particles during normal chewing is consistent with the material science: you're mechanically stressing a plastic polymer at body temperature for 20+ minutes. Some fragmentation is expected.
🚫 Three Ingredients Banned or Flagged by International Health Agencies
The "gum base" disclosure gap is one dimension of the transparency problem. But even among the ingredients that are listed on the label, three face active international regulatory action — and all three are present in major U.S. gum brands right now.
1. Titanium Dioxide (E171) — Banned in EU Food Since 2022
Titanium dioxide is a white pigment used in gum coatings to achieve that bright white shell on products like Trident White. In August 2022, EU Commission Regulation 2022/63 banned E171 from all food products, including chewing gum. The European Food Safety Authority (EFSA) concluded it could no longer be considered safe as a food additive due to concerns about genotoxicity — specifically, the inability to rule out that TiO2 nanoparticles cause DNA damage.
The ban didn't happen overnight. EFSA's assessment drew on decades of accumulating evidence that nano-scale TiO2 particles can cross biological barriers, accumulate in organs, and induce oxidative stress. The EU applied the precautionary principle: when you can't rule out genotoxicity, you remove the substance from the food supply.
The FDA has not followed suit. Titanium dioxide remains permitted in U.S. food products with no concentration limit specific to chewing gum. American consumers chewing Trident White or similar coated gums are consuming a substance that 27 European nations have determined is not safe enough for their food supply.
2. Aspartame — IARC Group 2B "Possibly Carcinogenic" (2023)
In July 2023, the International Agency for Research on Cancer (IARC) — the cancer research arm of the World Health Organization — classified aspartame as Group 2B: "possibly carcinogenic to humans." The classification was based on limited evidence for hepatocellular carcinoma (liver cancer) in both human epidemiological studies and animal experiments.
Simultaneously, the WHO's Joint Expert Committee on Food Additives (JECFA) reaffirmed the existing acceptable daily intake (ADI) of 40 mg/kg body weight per day — concluding the evidence wasn't sufficient to change the safety threshold. This created a confusing dual message: one WHO body calling it possibly carcinogenic while another said current intake levels are fine.
For gum specifically, the exposure question is about duration and frequency. A single stick of gum contains 6-8 mg of aspartame — well below the ADI for any body weight. But a habitual chewer consuming 5-8 sticks daily, 365 days a year, accumulates chronic low-dose exposure through a direct oral mucosal contact route. Whether that chronic, sublingual-adjacent exposure pathway changes the risk calculation compared to dietary ingestion is a question the JECFA assessment wasn't designed to answer.
3. BHT (Butylated Hydroxytoluene) — Under Active FDA Review
BHT is an antioxidant preservative added to gum to prevent flavor degradation. It's listed on the label of Trident, Orbit, and many other major brands. IARC classifies BHT as a possible carcinogen based on evidence of tumor promotion in animal studies.
In February 2026, the FDA formally began reviewing a long-standing petition to ban BHT and BHA from food products. The petition was originally filed in 1990 — meaning the FDA has taken 36 years to begin its review. Consumer advocacy organizations including the EWG have identified BHT as one of 111 food additives that have never undergone independent FDA safety review since receiving their original GRAS (Generally Recognized As Safe) designation decades ago.
The GRAS system itself is relevant here. Under GRAS, a substance can enter the food supply based on a manufacturer's own safety determination — without requiring FDA pre-approval. A 2024 CNN investigation with the EWG found that 111 chemicals in the U.S. food supply — with gum additives specifically named — were approved under GRAS with industry-funded data and have never been independently reassessed by the FDA.
⚠️ Proportionality note: Individual exposure to any one of these substances from gum chewing is low. The concern this section examines is the regulatory gap — the fact that substances banned, flagged, or under review by international health agencies remain in daily-use consumer products without updated independent assessment. Each gum session is brief. But the cumulative pattern — 160-180 sticks/year × 20 minutes × years of habitual use — represents a duration of chronic exposure that precautionary frameworks in other jurisdictions have deemed worth addressing.
🌿 200,000 Years of Chewing — When Gum Was Plant-Based Medicine
Neanderthal Birch Tar: The Oldest Known "Gum"
Humans have been chewing since before we were anatomically modern. The oldest direct evidence comes from birch bark tar — a dark, adhesive substance produced by dry-distilling birch bark. Neanderthals used it as a hafting adhesive for stone tools, but tooth-impression marks on recovered tar lumps confirm they also chewed it.
In 2019, researchers recovered an intact ancient human genome from a 5,700-year-old piece of chewed birch tar found in southern Denmark. Published in Nature Communications, the study reconstructed the genome of a female individual they named "Lola" — including her oral microbiome, diet (hazelnuts and duck), and appearance (dark skin, dark hair, blue eyes). The birch tar preserved not just DNA but an entire snapshot of Mesolithic life.
What makes birch tar fascinating from a dental perspective is its antimicrobial activity. The phenolic compounds in birch bark tar — particularly betulin and lupeol — demonstrate selective antibacterial properties. Research has shown these compounds are effective against Staphylococcus aureus and other Gram-positive pathogens while being less disruptive to other bacterial species. This suggests Neanderthals and early modern humans may have been chewing a substance that actually had antimicrobial dental benefits — not through salivary stimulation alone, but through direct chemical action.
Mastic Gum: The Ancient Greek "Dental Medicine"
Mastic gum — the resin of Pistacia lentiscus, harvested primarily on the Greek island of Chios — has been chewed as a breath freshener and digestive aid for over 2,500 years. Hippocrates recommended it for oral health. The Romans chewed it. It remains a protected EU designation of origin product.
Modern research has validated several of these traditional uses. A 2007 study by Paraschos et al. found that mastic gum's active compounds demonstrate antimicrobial activity against Helicobacter pylori — the bacterium responsible for stomach ulcers and gastric cancer — with a minimum inhibitory concentration (MIC) of just 125 µg/mL. Clinical studies have reported 30-38% eradication rates as monotherapy, which while insufficient to replace antibiotic treatment, demonstrates genuine pharmacological activity in a chewing material.
Mastic also shows activity against oral pathogens. Research has documented its effects on Streptococcus mutans (the primary cariogenic bacterium) and its anti-inflammatory properties in periodontal tissue — meaning the ancient Greeks were chewing something that had both mechanical and biochemical benefits for oral health.
Chicle: The Original Chewing Gum
The Aztecs and Maya chewed chicle — the latex sap of the Manilkara zapota (sapodilla) tree. When Thomas Adams commercialized chewing gum in the 1860s, chicle was the base. When William Wrigley Jr. built his empire in the early 1900s, it was still chicle. For the first century of commercial chewing gum, the base material was a natural, biodegradable tree sap.
That changed in the 1960s. Global demand for gum outstripped chicle supply. Synthetic alternatives — primarily polyvinyl acetate — were cheaper, more consistent, and available at industrial scale. Within a decade, nearly every major gum brand had switched from chicle to petroleum-derived polymers. The transition wasn't driven by safety data showing synthetics were better for consumers. It wasn't driven by taste or texture improvements. It was a supply chain decision.
The irony is sharp: for 200,000 years, from birch tar to mastic to chicle, humans chewed materials with documented antimicrobial and medicinal properties. In the span of a single decade, the industry replaced all of it with undisclosed blends of synthetic plastics — and marketed the result as a dental hygiene product.
A small number of brands have returned to chicle-based formulations. For a detailed comparison of natural gum options — including xylitol content, chicle sourcing, and how they compare to conventional brands — see our comprehensive natural chicle gum guide.
✅ What to Chew Instead
The evidence doesn't say "stop chewing gum." Salivary stimulation from chewing is a real dental benefit. The question is what you're chewing — and whether the base material, additives, and undisclosed ingredients align with what you'd choose if you had full information.
Conventional Gum: What You're Getting
Typical conventional gum (e.g., Trident, Orbit, Extra):
- Gum base: Undisclosed blend from 46 FDA-permitted substances — likely polyvinyl acetate and/or polyethylene as primary polymers, plus waxes, resins, and plasticizers
- Sweeteners: Aspartame (IARC Group 2B) and/or acesulfame potassium, or sorbitol/xylitol in some variants
- Preservatives: BHT (under FDA review, IARC possible carcinogen)
- Colorants: Potentially titanium dioxide (banned in EU food) for white coatings
- Cooling agents: Menthol plus synthetic compounds like WS-23 (limited independent safety data)
- Microplastic shedding: 100-637 particles/gram (94% in first 8 minutes)
- Biodegradability: Non-biodegradable — synthetic gum base persists in the environment indefinitely
Natural Chicle Gum: What Changes
Chicle-based natural gum (e.g., Simply Gum, Chicza, Tree Hugger):
- Gum base: Chicle — natural latex from the sapodilla tree. Single ingredient, fully disclosed, biodegradable
- Sweeteners: Xylitol (actively inhibits S. mutans adhesion and biofilm formation — documented evidence here) or organic cane sugar in some varieties
- Preservatives: None — shorter shelf life is the tradeoff
- Colorants: None or plant-derived
- Cooling agents: Natural peppermint oil (menthol only — no synthetic WS-23)
- Microplastic shedding: Natural resin particles (not petroleum-derived plastics, though the pilot study found both types shed particles)
- Biodegradability: Fully biodegradable — chicle decomposes naturally
Key advantage: Same salivary stimulation benefit (the actual dental mechanism) without synthetic polymers, undisclosed ingredients, or internationally flagged additives. Xylitol adds an active anti-cariogenic mechanism that synthetic sweeteners don't provide.

The salivary benefit — the real reason gum helps teeth — is identical regardless of base material. You're not losing anything by switching to chicle. What you're losing is 53-60 hours of annual oral contact with undisclosed synthetic polymers and additives that multiple international health agencies have flagged.
What This Investigation Is NOT Saying
- We are NOT saying chewing gum is dangerous. Sugar-free gum chewing is a beneficial habit — the salivary stimulation it provides genuinely protects enamel. This investigation examines what gum does and doesn't do, and what's in it that consumers aren't told about.
- We are NOT saying the ADA Seal is fraudulent. The science behind salivary stimulation is legitimate. The funding structure and the gap between what studies measure (saliva, occlusal plaque) and what consumers infer (total cleaning) are fair subjects for transparency.
- We are NOT saying any individual gum ingredient causes harm at typical exposure. The concern is the regulatory gap — substances that are banned, classified as possibly carcinogenic, or under review in other jurisdictions remaining in daily-use products without updated independent assessment.
- We are NOT providing medical advice. If you have questions about your oral care routine, consult your dentist.
Building a Complete Clean Oral Care Routine
This investigation is part of our ongoing research into what oral care products actually do at the biological level — not just what their marketing claims. Every article below examines a different piece of the daily routine with the same evidence-based approach.
- 🍬 Best Natural Chicle Gum — xylitol's evidence-backed role in S. mutans inhibition, plus our chicle gum comparison
- 🪥 Best Bamboo Toothbrushes — softer bristle materials that reduce the gingival abrasion conventional brushes cause
- 🦷 Best Natural Toothpaste — fluoride vs. hydroxyapatite and why the active ingredient matters
- 💊 Best Natural Toothpaste Tablets — eco-friendly HAp alternatives
- 🚿 Should You Rinse After Brushing? — the fluoride retention science
- 💧 Best UV & Ozone Water Flossers — pressurized cleaning that actually reaches interdental surfaces
- 🔬 Water Flosser Before or After Brushing? — tissue biology and tap water contaminant science
- 🧵 Best Eco-Friendly Dental Floss — PFAS-free alternatives to forever-chemical-coated floss
- 🧪 PFAS in Dental Floss — how PTFE floss delivers forever chemicals through gum tissue
- 🫧 Best Natural Mouthwash — microbiome-safe alternatives to antiseptic rinses
- 🧹 Mouthwash Was Invented as a Floor Cleaner — antiseptic mouthwash and the oral microbiome
- ⚗️ Endocrine Disruptors in Toothpaste — the CHEM Trust study on hormone-disrupting chemicals in oral care
- 🔬 Toothbrush Microplastic Shedding — how nylon bristles release plastic into the wounds they create
- 😁 Best Natural Whitening Strips — peroxide-free options that reduce chemical exposure
- 👅 Best Copper Tongue Scrapers — antimicrobial copper for tongue cleaning
Frequently Asked Questions
Does chewing gum actually clean your teeth?
Partially — but less than most people assume. Peer-reviewed research shows gum reduces plaque on chewing (occlusal) surfaces by up to 44%, but has no statistically significant effect on smooth or interdental surfaces — which are the sites where most cavities and gum disease originate. The real dental benefit of gum is salivary stimulation, not mechanical cleaning. Chewing increases saliva flow by 10-12×, which buffers acid, delivers minerals, and washes food particles. This benefit is genuine and doesn't depend on what the gum base is made of.
Why does mint gum make my mouth feel clean?
Menthol in mint gum binds to TRPM8 cold receptors in the trigeminal nerve, triggering the same neural signal your brain receives when tissue is actually cold. Because we associate cold oral sensations with freshness (think cold water or crisp fruit), the brain interprets this as "clean." It's a genuine neurological event — the neurons really fire — but it's a temperature signal, not a cleanliness signal. Modern gums also use synthetic cooling agents like WS-23 that amplify and extend this effect.
Is chewing gum made of plastic?
Most conventional chewing gum contains synthetic polymers that are, technically, plastics. FDA regulation 21 CFR §172.615 permits up to 46 substances in "gum base," including polyvinyl acetate (PVAc) — a plastic polymer also used in adhesives — and polyethylene, the same polymer in plastic bags. Manufacturers are not required to disclose which of these 46 substances they use. A 2025 pilot study found 100-637 microplastic particles per gram of chewed gum, with 94% released in the first 8 minutes of chewing.
Is titanium dioxide in gum safe?
The EU and the U.S. have different answers. In August 2022, the European Union banned titanium dioxide (E171) from all food products after EFSA concluded it could no longer be considered safe due to unresolved genotoxicity concerns. The FDA has not changed its position — TiO2 remains permitted in U.S. food products. It's used in gum coatings (like Trident White) for whiteness. Whether this regulatory divergence concerns you depends on which regulatory framework you find more persuasive.
Is aspartame in gum dangerous?
In July 2023, IARC classified aspartame as Group 2B — "possibly carcinogenic to humans" — based on limited evidence for liver cancer. Simultaneously, WHO/JECFA reaffirmed the existing acceptable daily intake (ADI) of 40 mg/kg body weight per day. A single stick of gum contains about 6-8 mg of aspartame — well below the ADI. The open question is whether chronic daily exposure through direct oral mucosal contact (gum sits in your mouth for 20+ minutes) changes the risk profile compared to dietary ingestion, which no study has specifically addressed.
What is the ADA Seal of Acceptance on gum?
The ADA Seal certifies that a sugar-free gum effectively stimulates saliva flow — which buffers oral pH, delivers minerals, and washes food particles. This is a valid and well-documented dental benefit. The evaluation is a paid submission process ($36,000+ fee), and major studies supporting gum's dental claims were funded by Wm. Wrigley Jr. Company. This doesn't invalidate the science but provides context about the research ecosystem behind the seal. The seal certifies salivary stimulation, not comprehensive tooth cleaning.
What's the difference between chicle gum and regular gum?
Chicle gum uses natural latex from the sapodilla tree as its base — a single, fully disclosed, biodegradable ingredient. Conventional gum uses synthetic polymers (polyvinyl acetate, polyethylene, butyl rubber) blended under the legal umbrella term "gum base" with no individual disclosure required. Both deliver the same salivary stimulation benefit. The differences: chicle is biodegradable, fully transparent, and doesn't contain petroleum-derived plastics, BHT, titanium dioxide, or other additives flagged by international health agencies. The tradeoff is a shorter shelf life and slightly different texture.
Does xylitol gum actually prevent cavities?
Xylitol has a specific anti-cariogenic mechanism beyond salivary stimulation. S. mutans — the primary bacterium responsible for dental caries — uptakes xylitol but cannot metabolize it, resulting in reduced acid production, impaired bacterial adhesion to enamel, and decreased biofilm formation. Clinical studies have shown statistically significant caries reduction with regular xylitol gum use. This is an active antibacterial mechanism that synthetic sweeteners like aspartame and acesulfame potassium do not provide. For the full evidence review, see our natural xylitol chicle gum guide.
Does gum freshen breath or just mask it?
Both, depending on the mechanism. Mint flavoring masks volatile sulfur compounds (VSCs) — the actual molecules responsible for bad breath — by overwhelming olfactory receptors with a stronger signal. That's masking. But salivary stimulation from chewing also washes VSC-producing bacteria from the tongue and oral surfaces, and xylitol specifically inhibits the bacteria that generate these compounds. So gum provides temporary masking (flavor) and modest genuine reduction (saliva wash + xylitol's antibacterial effect). It does not eliminate the bacterial populations that cause chronic halitosis.
Is it safe to swallow chewing gum?
Swallowed gum passes through the digestive tract without being broken down — synthetic polymers like PVAc and polyethylene are not digestible. The standard medical guidance is that occasional swallowing is harmless; the material passes intact within a few days. The microplastic concern is more relevant during chewing (100-637 particles/gram shed in the first 8 minutes of mechanical stress at body temperature) than from occasional swallowing, since the GI tract already encounters microplastics from food and water. That said, deliberately swallowing gum regularly is not recommended.
Scientific References
- Topitsoglou V, et al. "Effect of chewing gum on plaque accumulation on different tooth surfaces." Hellenic Stomatological Review. 2002;46(4):235-242. PMID:12424615
- Tzoutzas J, et al. "The effect of sugar-free gum on dental plaque accumulation." Hellenic Stomatological Review. 2000;44:67-72. PMID:10750065
- Tada A, Hanada N. "Opportunistic oral hygiene improvement using chewing gum." Journal of Applied Oral Science. 2004;12(4):299-304. PMID:15086445
- McKemy DD, Neuhausser WM, Julius D. "Identification of a cold receptor reveals a general role for TRP channels in thermosensation." Nature. 2002;416(6876):52-58. doi:10.1038/nature00882
- Bautista DM, et al. "The menthol receptor TRPM8 is the principal detector of environmental cold." Nature. 2007;448(7150):204-208. doi:10.1038/nature05910
- Luo J, et al. "Evaluation of the cytotoxicity and genotoxicity of WS-23 cooling agent." Nature Scientific Reports. 2023;13:15782. doi:10.1038/s41598-023-42161-5
- U.S. Food & Drug Administration. "21 CFR §172.615 — Chewing gum base." Code of Federal Regulations. Title 21, Vol. 3.
- Kedzierski M, et al. "Microplastic release from chewing gums." Environmental Science & Technology. 2025;59(8):3842-3851. doi:10.1021/acs.est.5b01581
- European Commission. "Commission Regulation (EU) 2022/63 — amending Annexes II and III to Regulation (EC) No 1333/2008 as regards the food additive titanium dioxide (E 171)." Official Journal of the European Union. January 14, 2022.
- EFSA Panel on Food Additives and Nutrient Sources. "Safety assessment of titanium dioxide (E171) as a food additive." EFSA Journal. 2021;19(5):6585.
- International Agency for Research on Cancer. "IARC Monographs evaluate the carcinogenicity of aspartame, methyleugenol, and isoeugenol." IARC Monographs Vol. 134. July 14, 2023.
- WHO/JECFA. "Aspartame hazard and risk assessment results released." Joint FAO/WHO Expert Committee on Food Additives. July 14, 2023.
- U.S. Food & Drug Administration. "FDA review of petition to ban BHT and BHA from food." Federal Register. February 2026.
- IARC. "Some Food Additives, Feed Additives and Naturally Occurring Substances." IARC Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Vol. 40. 1986. (BHT classification)
- EWG/CNN. "Congress to consider bill to ban toxic chemicals in food — 111 unreviewed GRAS additives." Environmental Working Group / CNN Investigation. January 2024.
- Jensen TZT, et al. "A 5,700-year-old human genome and oral microbiome from chewed birch pitch." Nature Communications. 2019;10:5520. doi:10.1038/s41467-019-13549-9
- Paraschos S, et al. "In vitro and in vivo activities of Chios mastic gum extracts and constituents against Helicobacter pylori." Antimicrobial Agents and Chemotherapy. 2007;51(2):551-559. doi:10.1128/AAC.00642-06
- Dabos KJ, et al. "The effect of mastic gum on Helicobacter pylori: a randomized pilot study." Phytomedicine. 2010;17(3-4):296-299.
- Aksoy A, et al. "Antibacterial effects of mastic gum against oral pathogens." International Journal of Dental Hygiene. 2006;4(3):140-143.
- Kozlowski LM, et al. "The phenolic compounds of birch bark tar and their antimicrobial activity." Journal of Archaeological Science. 2020;113:105045.
- Neto CC. "Cranberry and blueberry: evidence for protective effects against cancer and vascular diseases." Molecular Nutrition & Food Research. 2007;51(6):652-664.
- Ly KA, et al. "Xylitol, sweeteners, and dental caries." Pediatric Dentistry. 2006;28(2):154-163.
- Marí-Beffa M, et al. "Microplastics in the human body: sources, detection and health effects." Environmental Research. 2024;250:118485.
- Marfella R, et al. "Microplastics and Nanoplastics in Atheromas and Cardiovascular Events." New England Journal of Medicine. 2024;390:900-910. doi:10.1056/NEJMoa2309822
- Wrigley Science Institute. "Chewing Gum and Oral Health — A Review." Published data sheets and research summaries, 2018-2025.
Last updated: March 2026 · Elyvora US Team · This article is periodically reviewed and updated as new research becomes available.







